What Is a Proton Exchange Membrane Fuel Cell
A PEM fuel cell (Proton Exchange Membrane fuel cell) — also called a Polymer Electrolyte Membrane fuel cell — is an electrochemical device that converts the chemical energy of hydrogen and oxygen directly into electricity, producing only water and heat as byproducts. Operating at relatively low temperatures of 60-80°C, PEM fuel cells achieve 40-60% electrical efficiency — significantly higher than the 20-35% efficiency of combustion engines.
Used in hydrogen fuel cell electric vehicles (FCEVs) like the Toyota Mirai, Hyundai Nexo, and hydrogen-powered forklifts (Plug Power supplies ~30% of US warehouse food handling), as well as backup power for telecom and data centers, PEM fuel cells offer quick startup, scalability from kW to MW, and zero tailpipe emissions. Their compact, modular design makes them the leading fuel cell type for transportation, portable power, and stationary backup applications in the emerging hydrogen economy.
How Does a PEM Fuel Cell Work
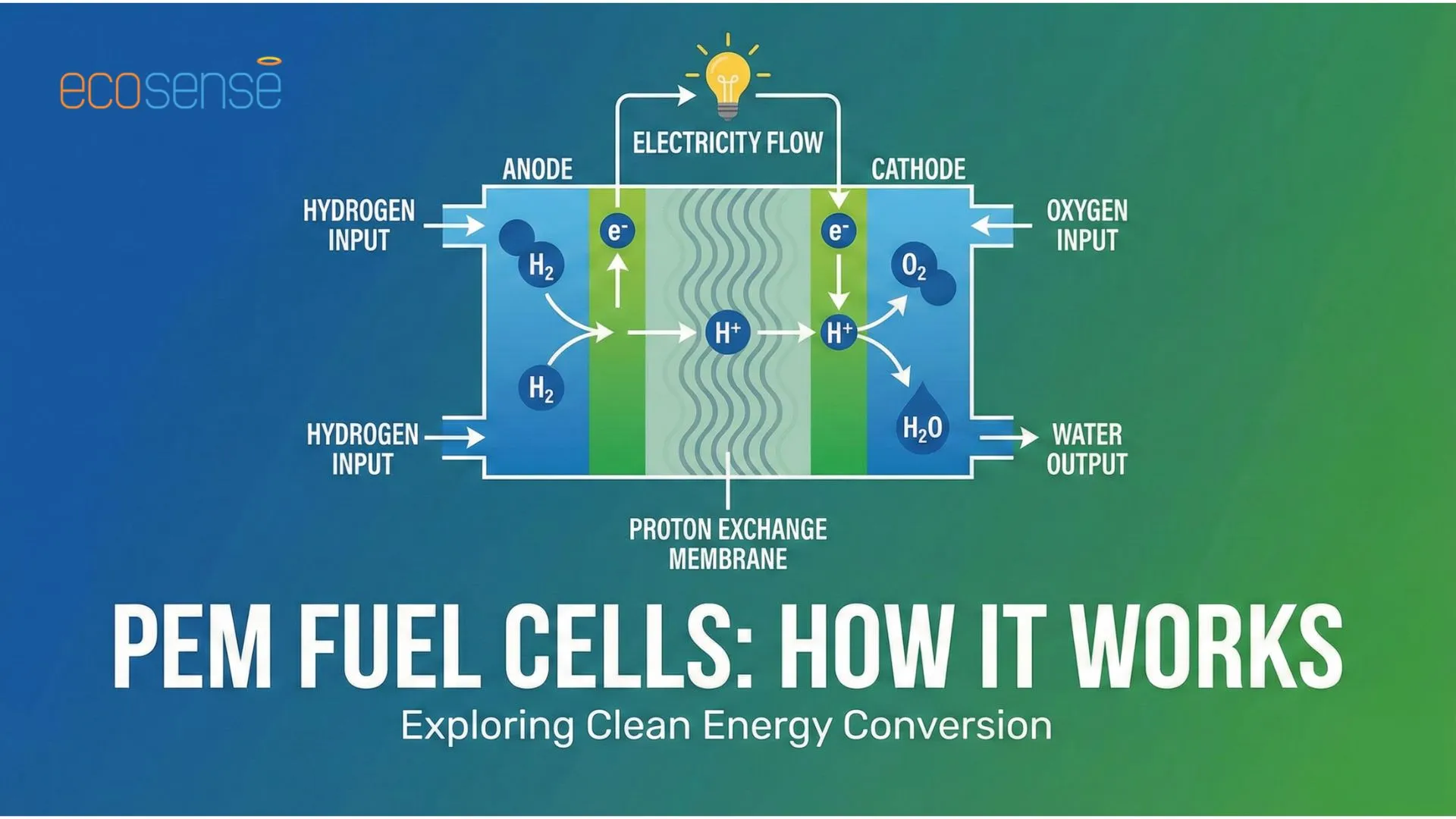
In a polymer electrolyte membrane fuel cell, hydrogen is supplied to the anode and oxygen is supplied to the cathode. The system uses a catalyst, usually platinum, to accelerate the reactions.
At the anode, hydrogen molecules split into protons and electrons. The pem fuel cell membrane allows only protons to pass through it, while electrons are forced to travel through an external circuit. This movement of electrons generates electrical current.
At the cathode, oxygen reacts with the incoming protons and electrons to form water and heat.
2H₂(g) + O₂(g) → 2H₂O(l)
Anode (Oxidation): Hydrogen gas loses electrons (is oxidized).
H₂(g) → 2H⁺(aq) + 2e⁻ (Acidic Medium)
Cathode (Reduction): Oxygen gas gains electrons (is reduced).
½O₂(g) + 2H⁺(aq) + 2e⁻ → H₂O(l) (Acidic Medium)
The overall process is called a redox reaction where simultaneous oxidation- loss of electrons at the anode and reduction - gain of electrons at the cathode occurs. These reactions are generally slow, so there is a need for catalysts such as platinum to speed up the reaction to give up electrical energy with water and heat as the by-product. This process makes the polymer electrolyte membrane fuel cell highly efficient and environmentally friendly.
Fig. How PEM Fuel Cell Works
- Role of Anode: Hydrogen is supplied to anode. Anode facilitates hydrogen oxidation and releases electrons to the external circuit as well as supplies protons to the membrane
- Role of Proton Exchange Membrane: The membrane is solid acidic polymer typically made from perfluorosulfonic acid (PFSA) polymers such as Nafion. It only allows protons to pass from anode to cathode and blocks electron forcing them through an external circuit creating current. It also prevents direct mixing of hydrogen and oxygen.
- Role of Cathode: Oxygen is supplied to the cathode. Oxygen reacts with incoming protons and electrons to form water and heat to complete electrochemical circuit.
Components of a PEM Fuel Cell
A PEM fuel cell is built from a stack of carefully engineered components, each playing a specific role in the electrochemical conversion process:
Proton Exchange Membrane (PEM)
Thin polymer electrolyte sheet (typically Nafion or PFSA). Allows H⁺ protons to pass while blocking electrons. Requires >10⁻² S/cm proton conductivity.
Membrane Electrode Assembly (MEA)
The functional core: PEM + catalyst layers + gas diffusion layers + gaskets bonded together. Determines power density and efficiency of the cell.
Catalyst Layers
Platinum-based nanoparticles (or Pt-alloys like Pt-Co) on anode and cathode. Accelerate hydrogen oxidation (HOR) and oxygen reduction (ORR) reactions.
Bipolar Plates
Stacked conductive graphite or metal plates. Distribute H₂/O₂ gases via flow channels, conduct electricity between cells, manage cooling water.
Gas Diffusion Layers (GDL)
Porous carbon paper between catalyst and bipolar plates. Ensures uniform gas distribution to catalyst sites and manages water transport.
Gaskets & Seals
Gas-tight rubber/silicone seals around MEA edges. Prevent leakage of hydrogen, oxygen, water, and coolant between cells in the stack.
PEM vs Other Fuel Cell Types
PEM is one of five major fuel cell technologies, each suited to different operating temperatures, fuel sources, and applications:
PEM Fuel Cell
Efficiency: 40–60%
Fuel: Pure H₂
Best for: Transport, portable
Quick startup, compact, scalable from W to MW. Dominates FCEVs and material handling.
Solid Oxide (SOFC)
Efficiency: 50–70%
Fuel: H₂, NG, biogas
Best for: Stationary CHP
Highest efficiency. Fuel-flexible (no need for pure H₂). Used by Bloom Energy for data centers.
Alkaline (AFC)
Efficiency: 60–70%
Fuel: Pure H₂ + Pure O₂
Best for: Aerospace
Used in Apollo and Space Shuttle. Highly sensitive to CO₂ contamination — limits commercial use.
Phosphoric Acid (PAFC)
Efficiency: 36–42%
Fuel: H₂, natural gas
Best for: Stationary CHP
First fuel cell commercialised. Used in hospital backup power and telecom installations.
Molten Carbonate (MCFC)
Efficiency: 50–60%
Fuel: H₂, NG, biogas, coal
Best for: Utility power
Multi-MW utility plants. Internal reforming of fuels. FuelCell Energy is a major commercial supplier.
Detailed Comparison Table:
Applications of PEM Fuel Cells
PEM fuel cells' low operating temperature, quick startup, and compact design make them suitable for a wide range of applications across transportation, stationary power, and portable electronics:
FCEVs (Cars)
Toyota Mirai, Hyundai Nexo, Honda Clarity. ~3 min refueling time
Buses & Trucks
Hydrogen city buses in California, Japan, Europe. Long-range heavy-duty
Material Handling
Plug Power forklifts power ~30% of US warehouse food handling
Telecom Backup
Plug Power GenSure for cell tower & remote backup power
Data Center Backup
Backup power for high-availability data centers replacing diesel gensets
Hydrogen Trains
Coradia iLint (Alstom) — first hydrogen passenger train in regular service
Marine & Ferries
Hydrogen-powered ferries piloting in Norway, Netherlands, San Francisco
Portable Electronics
Laptops, drones, military comms — Samsung SDI 15-hour laptop fuel cell
Key Advantages of PEM Fuel Cells
- High Power Density and Fast Startup: Polymer Electrolyte Membrane fuel cells can deliver a lot of power from a relatively small system. They start up quickly and respond almost instantly to changes in power demand, which is why they work so well in vehicles and other applications where quick performance matters.
- Low Operating temperature and compact design: Because Polymer Electrolyte Membrane fuel cells operate at low temperatures, they don’t need long warm-up times or bulky thermal systems. This allows the overall system to stay compact, lightweight, and easier to integrate into real-world and laboratory setups.
- Zero local emission and Quiet Operation: PEM fuel cells generate electricity without combustion, producing only water and heat as by-products. This means no local pollution and very quiet operation, making them ideal for indoor environments, urban mobility, and hands-on learning labs.
Common Uses of PEM Fuel Cell
- Transportation and Mobility: The polymer electrolyte membrane fuel cell is widely used in electric vehicles, buses, and hydrogen-powered mobility systems. Its ability to deliver instant power and operate efficiently makes it a strong alternative to internal combustion engines.
- Stationary Power Systems: In stationary applications, the polymer electrolyte membrane fuel cell provides reliable backup power for critical infrastructure such as hospitals, telecom towers, and data centers.
- Portable and Research Applications: Compact systems based on polymer electrolyte membrane fuel cell technology are used in portable devices, drones, and laboratory experiments. These systems are ideal for controlled testing and educational purposes.
Limitations and Challenges of PEM Fuel Cell
- High Material Cost: One of the biggest challenges is the high proton exchange membrane cost. The membrane material and platinum catalyst significantly increase the overall system cost. Reducing the proton exchange membrane cost is essential for large-scale commercialization of the polymer electrolyte membrane fuel cell. Researchers are actively working on alternative materials and cost-effective manufacturing methods.
- Fuel Purity Requirements: The polymer electrolyte membrane fuel cell requires very high-purity hydrogen for efficient operation. Even small impurities can affect performance and damage the system.
- Water Management: Maintaining proper hydration of the pem fuel cell membrane is critical. Excess water can flood the system, while insufficient hydration can dry out the membrane and reduce efficiency.
- Durability: Over time, the pem fuel cell membrane and catalyst layers degrade due to thermal and chemical stress. Improving durability remains a key focus area.
Future Scope
The polymer electrolyte membrane fuel cell is expected to play a major role in the transition to clean energy. Advances in materials, catalyst reduction, and system design are helping improve efficiency and reduce costs.
Efforts to lower the proton exchange membrane cost and enhance membrane durability are making the technology more viable for widespread adoption. With continued innovation, the polymer electrolyte membrane fuel cell will become increasingly important in transportation, grid systems, and hydrogen-based energy solutions.
PEM Fuel Cells in India
India's National Green Hydrogen Mission (NGHM)— approved January 2023 with ₹19,744 crore allocation — is positioning PEM fuel cells as a cornerstone technology for the country's clean transportation and industrial decarbonization:
- Target: 5 million metric tonnes of green hydrogen production annually by 2030
- SIGHT scheme: Strategic Interventions for Green Hydrogen Transition — ₹17,490 crore for production incentives
- Pilot projects: ₹1,466 crore allocated for hydrogen-vehicle and shipping pilots, including PEM fuel cell trials
- R&D: ₹400 crore for fuel cell + electrolyser research at Indian universities and DRDO
- Tata Motors hydrogen buses: 15 hydrogen-powered city buses operational in pilot fleets
- Indian Oil Corporation (IOCL): Operating hydrogen fueling station pilots in Faridabad and Delhi
- Reliance Industries: Major Green Hydrogen + electrolyzer + fuel cell ecosystem investments announced 2023-2024
- Adani New Industries: Hydrogen mobility and PEM fuel cell stack development
- BHEL (Bharat Heavy Electricals): Indigenous PEM fuel cell stack development for stationary power
- Indian Railways: Trial hydrogen trains on Jind-Sonipat route planned for 2025-2026
Conclusion
The polymer electrolyte membrane fuel cell is a highly efficient and clean energy technology that offers significant advantages over traditional power systems. Its fast startup, compact design, and zero emissions make it suitable for a wide range of applications.
However, challenges such as high proton exchange membrane cost, strict fuel purity requirements, and durability issues still need to be addressed. With ongoing research and technological advancements, the polymer electrolyte membrane fuel cell is set to become a key component of the future energy landscape.